流动海水不仅加速溶解氧向管路表面扩散速率,也会导致防护性腐蚀产物膜剥落,从而加速电偶腐蚀[13-15]。国内外学者对于不同管路材料之间的电偶腐蚀做过大量研究,戴明安等[16,17]利用电化学以及失重法研究了945钢与铜锌合金、钛合金、不锈钢和锌基牺牲阳极4种偶对在0~11 m/s的流动海水中的电偶腐蚀行为,结果表明,流速越高,电偶腐蚀作用越大。孙保库等[18]通过腐蚀电位监测、电偶腐蚀实验考察了海水流速对B10和H62两种材料电偶腐蚀倾向、电偶腐蚀规律的影响,表明在不同流速海水中,H62黄铜与B10间存在明显的电偶腐蚀倾向,随海水流速增大,电偶腐蚀效应先减小后增大。王健等[19]研究了316L/2205和431/2205偶对在3.5% (质量分数) NaC1溶液中电偶腐蚀行为,表明介质流动显著增大电偶电流。
1 实验方法
实验材料为20#钢和ZCuSn5Pb5Zn5合金,试样规格为Ф50×20 mm,电偶对面积比1∶1。20#钢化学成分 (质量分数,%) 为Cr 0.183,Mo 0.011,Co 0.057,Cu 0.042,Mn 0.433,Bi 0.197,Fe余量。ZCuSn5Pb5Zn5合金的化学成分 (质量分数,%) 为Sn 4.735,Pb 5.165,Zn 4.944,S 0.405,Zr 0.025,Hg 0.64,Cu余量。
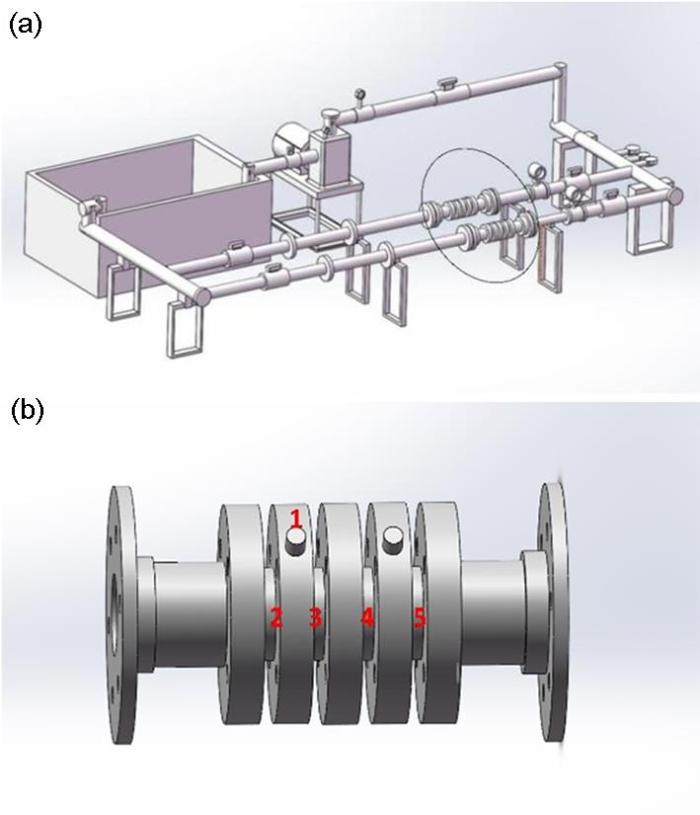
20#钢/ZCuSn5Pb5Zn5合金电偶冲刷腐蚀实验在中国船舶集团公司第七二五研究所自主设计研制的海水管路综合模拟平台进行,该平台可对海水流速进行监控,并实时测量海水温度、压力、pH值和电导率。管路冲刷装置如图1a所示,其中椭圆形标记处为测试段,测试段的电解池装置如图1b所示,电解池装置的1处为参比电极安装位置,2~5处为管状20#钢和ZCuSn5Pb5Zn5合金试样以及钛基贵金属氧化物阳极辅助电极安装位置。试样、辅助电极、电解池及冲刷试验装置管径相同,且试验装置前后直管长度达到20倍管径长度,确保海水以恒定的速度流经试样、辅助电极和参比电极表面,避免试样表面形成湍流和起泡影响测试结果准确性。本次实验测试温度为14±1 ℃,实验介质为天然海水,取自青岛鳌山湾海域。待海水流速稳定后,使用CS353便携式电化学工作站测量电偶电位和电偶电流,20#钢接工作电极、ZCuSn5Pb5Zn5合金接地线,测试周期为8 h或16 h,电偶电位与电偶电流稳定后停止测量。
图1
图1
管路冲刷装置及电解池装置示意图
Fig.1
Schematic diagrams of pipeline scouring device (a) and electrolytic cell device (b)
20#钢/ZCuSn5Pb5Zn5合金电偶静态海水长期腐蚀试验方法按照GB/T 6384-2008开展,将试样投放至青岛鳌山湾海域。通过两块PVC板将管路材料固定在中间,然后将组装好的PVC板安装到钛合金框架上,管路试样与钛合金框架绝缘,使用自主研发的原位电化学测试装置测量管路试样电偶电位和电偶电流,参比电极为Ag/AgCl电极。
实验结束后利用KH8700数字视频显微镜 (OM)、ULTRA55场发射扫描电子显微镜观察腐蚀产物形貌;利用LabRAM HR EVOLUTION拉曼光谱仪 (激光拉曼光谱采用氩离子激光器,光束直径2 mm,镜头为100x长焦,激发波长533 nm,扫描范围100~2000) 分析腐蚀产物成分。
2 流速对电偶腐蚀速率影响试验研究
2.1 流速对腐蚀电位影响研究
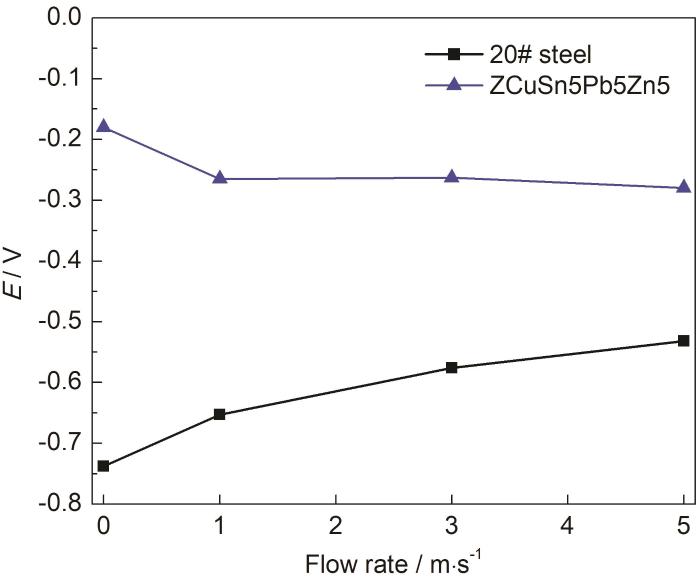
20#钢和ZCuSn5Pb5Zn5合金在静态以及1、3、5 m/s流速海水中的腐蚀电位至少进行144 h的连续监测测试,参比电极为Ag/AgCl电极,结果如图2所示。由测试结果可知,20#钢和ZCuSn5Pb5Zn5合金在不同海水流速下腐蚀电位差均在250mV以上;当20#钢和ZCuSn5Pb5Zn5合金偶接时,会产生严重的电偶腐蚀,20#钢将作为阳极被加速腐蚀,ZCuSn5Pb5Zn5合金则会作为阴极受到一定的保护。20#钢腐蚀电位随着流速增加而正移,而ZCuSn5Pb5Zn5合金腐蚀电位随着流速增加而减小,二者腐蚀电位差随着流速增加而减小,电偶腐蚀驱动电压随着流速增加而减小。
图2
图2
20#钢和ZCuSn5Pb5Zn5合金在不同流速海水中的平均腐蚀电位
Fig.2
Average corrosion potentials of 20# steel and ZCuSn5Pb5Zn5 alloy in seawater with different flow rates
2.2 静态海水环境电偶腐蚀实验
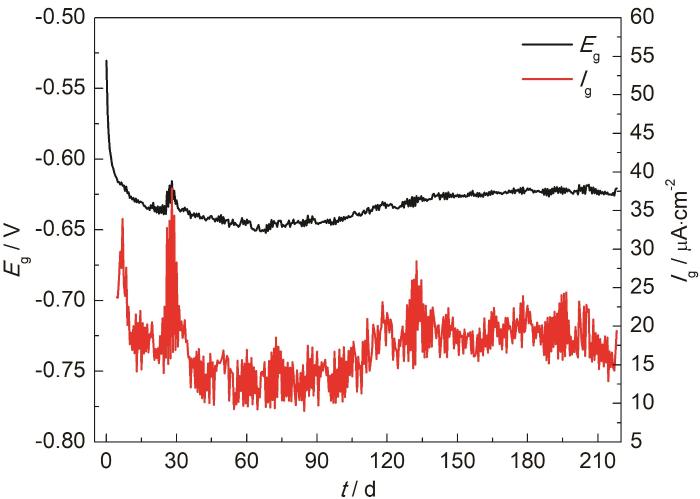
20#钢与ZCuSn5Pb5Zn5合金按面积比1∶1组成电偶对后,置于青岛鳌山湾海水中453 d。通过自主研发的原位电化学测试装置测量管路试样前218 d的电偶电位和电偶电流,测量结果如图3所示。由测量结果可知,20#钢/ZCuSn5Pb5Zn5合金偶对初始电偶电位为-530.3 mV,阳极极化过电位为151.2 mV,初始电偶电流23.75
图3
图3
20#钢/ZCuSn5Pb5Zn5合金偶对的电偶电位和电偶电流随时间的变化
Fig.3
Variations of galvanic potential and galvanic current of 20# steel/ZCuSn5Pb5Zn5 alloy couple with corrosion time
式中,v为腐蚀速率,mm/a;Ig为电偶电流密度,A/cm2;M为分子量,g/mol;F为Faraday常数,c/mol。
采用添加六次甲基四胺缓蚀剂的HCl酸洗液,去除20#钢试样表面腐蚀产物,通过失重法计算得到20#钢总腐蚀速率为0.27 mm/a,其在海水中的自腐蚀速率为0.10 mm/a,则电偶腐蚀速率为0.17 mm/a。对比电化学测试结果和失重试验结果可知,由于电化学测试时间较失重实验时间相对较短,且前期电偶腐蚀速率较大,因此,电化学测试结果稍大于长期失重实验结果。
2.3 流动海水环境电偶腐蚀试验
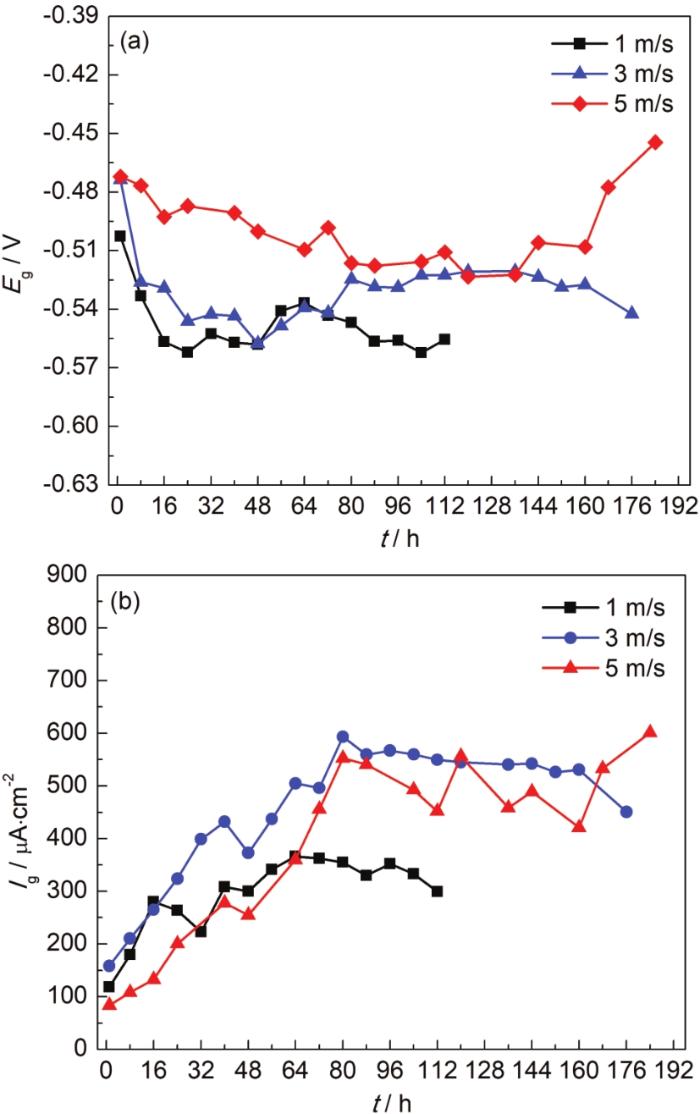
20#钢/ZCuSn5Pb5Zn5合金偶在不同流速海水中电偶电位测试结果如图4a所示。由测试结果可知,流速越大,20#钢电偶电位越正,说明电偶腐蚀倾向越大;冲刷初期电偶电位较正,20#钢电偶腐蚀倾向较大,随着冲刷时间增加,腐蚀产物逐渐沉积对电偶腐蚀起到抑制作用,电偶电位逐渐负移并趋于稳定。
图4
图4
20#钢/ZCuSn5Pb5Zn5合金偶在不同流速下的电偶电位及电偶电流密度随时间变化曲线
Fig.4
Galvanic potentials (a) and galvanic current densities (b) of 20# steel/ZCuSn5Pb5Zn alloy couple in seaw-ater with different flow rates
图4b为20#钢/ZCuSn5Pb5Zn5合金电偶在不同流速海水中电偶电流测试结果。随着冲刷的进行,电偶电流均呈现先迅速增长后小幅度下降并趋于稳定的趋势,这主要是因为在冲刷初期,20#钢表面金属溶解反应和ZCuSn5Pb5Zn5合金表面氧还原反应活性较强,在电偶效应作用下20#钢腐蚀电流急剧增大;随着阳极表面腐蚀产物层的形成,减小了电偶腐蚀活性面积,电偶电流密度下降并趋于稳定。
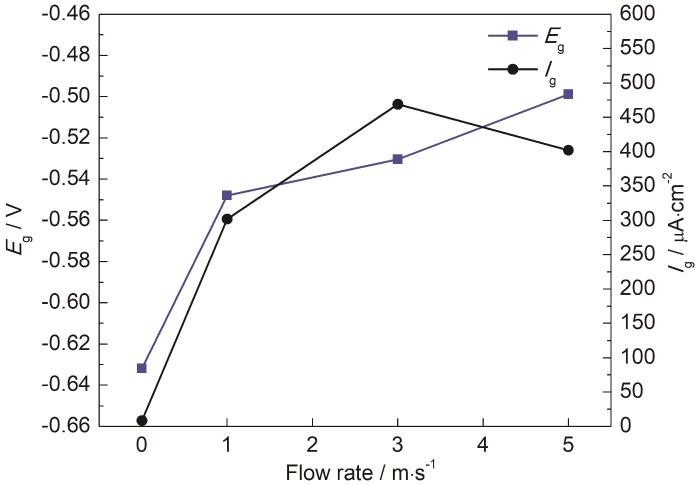
不同流速条件下20#钢/ZCuSn5Pb5Zn5合金平均电偶电位如图5所示。可知,平均电偶电位总体趋势呈现随着流速增加而正移。海水由静态变为流动状态后,电偶电位迅速正移至-550 mV以上;在流动海水中电偶电位处于-550~-500 mV范围内,电偶电位变化速率减小。由图5所示的不同流速条件下的电偶电流平均值可知,随着流速的升高,偶对电偶电流密度先显著增大后小幅度减小,偶对在1 m/s流速海水中的电偶电流密度是静态下的17.5倍,说明在流动海水作用下20#钢电偶腐蚀急剧加重。当海水流速从1 m/s增大到3 m/s时,偶对电偶电流密度升高至最大值469
图5
图5
电偶电流密度和电偶电位随流速变化曲线
Fig.5
Variation curves of galvanic current density and galvanic potential of the couple with the flow rate of seawater
2.4 腐蚀形貌及腐蚀产物成分分析
电偶腐蚀实验后,20#钢表面均附着有一层厚厚的腐蚀产物,内层为黑色腐蚀产物,外层为黄褐色腐蚀产物,如图6所示。这说明20#钢作为电偶对阳极,发生了严重的腐蚀。随着海水流速的增大,20#钢表面黄褐色的锈层面积逐渐减小,而黑色的内锈层面积逐渐增加,尤其在5m/s的流速下疏松的黄褐色锈层几乎被完全冲刷掉,主要以强附着力黑色腐蚀产物为主,腐蚀产物膜防护性能较好,可能是20#钢在5 m/s流动海水中电偶腐蚀速率较3 m/s流动海水中电偶腐蚀速率略有减小的重要原因。
图6
图6
偶接20#钢在不同流速冲刷后的腐蚀形貌
Fig.6
Corrosion morphologies of coupled 20# steel after scouring in seawater with the flow rates of 0 m/s (a), 1 m/s (b), 3 m/s (c) and 5 m/s (d)
20#钢去除腐蚀产物后放大三维形貌图如图7所示。由图可见,实验后20#钢表面凹凸不平,腐蚀严重,有大量的腐蚀坑产生。在1 m/s的流速下点蚀坑较浅,通过测量20#钢在1 m/s流速下的点蚀深度为59.008
图7
图7
偶接20#钢腐蚀并经酸洗去除产物膜后的三维表面形貌
Fig.7
3D morphologies of coupled 20# steel after corrosion in seawater with 1 m/s (a), 3 m/s (b) and 5 m/s (c) flow rates and then removing the surface corrosion products by pickling
20#钢腐蚀后微观形貌如图8所示。可以看出,在1 m/s流动海水中,20#钢表面腐蚀产物较多并且疏松多孔,腐蚀产物形状主要为棉絮状形貌。海水流速不低于3 m/s时,海水冲刷作用增强,表面疏松产物剥离,20#钢表面腐蚀产物形状主要为球形、针状形貌。
图8
图8
阳极20#钢在不同流速海水中腐蚀后的微观形貌
Fig.8
Micromorphologies of anodic 20# steel after corrosion in seawater with the flow rates of 1 m/s (a), 3 m/s (b) and 5 m/s (c)
图9
图9
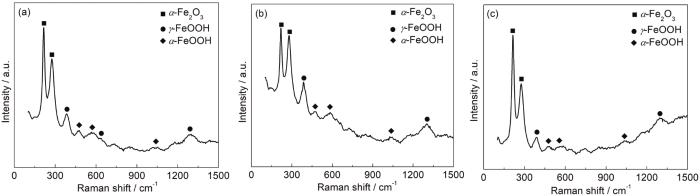
20#钢在不同流速海水中形成的表面腐蚀产物的拉曼光谱分析
Fig.9
Raman spectroscopies of surface corrosion products of 20# steel after corrosion in seawater with the flow rates of 1 m/s (a),3 m/s (b) and 5 m/s (c)
20#钢去除腐蚀产物后的微观形貌如图10所示,电偶腐蚀试验后20#钢表面凹凸不平,沟壑较深,腐蚀严重。20#钢作为阳极被加速腐蚀表面存在大量腐蚀坑,表现出点蚀加均匀腐蚀的特征;20#钢点蚀面积和点蚀深度均随着流速增加而增大。
图10
图10
阳极20#钢在不同流速海水中腐蚀并酸洗去除表面产物膜后的微观形貌
Fig.10
Micromorphologies of 20# steel after corrosion in seawater with 0 m/s (a), 1 m/s (b), 3 m/s (c) and 5 m/s (d) flow rates and then removing the corrosion products by pickling
3 20#钢/ZCuSn5Pb5Zn5电偶腐蚀机制分析
20#钢和ZCuSn5Pb5Zn5合金在海水中接触后,20#钢失去电子发生溶解,ZCuSn5Pb5Zn5合金阴极发生氧的还原,由于海水中的溶解氧致使腐蚀初始阶段形成的中间产物Fe(OH)2氧化生成FeOOH,FeOOH进一步反应形成了难以分解的Fe2O3。
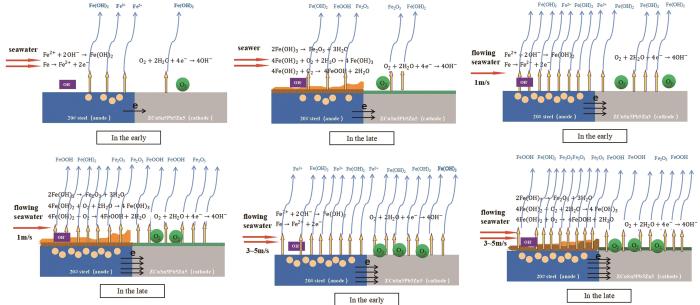
由图11极化曲线可知,无论是冲刷初期还是冲刷后期,与静态海水相比,由于流动海水使得溶解氧扩散速率增加,同时增强Cl-、OH-的传输能力,相同阳极极化电位下,20#钢在1 m/s流动海水中阳极极化电流密度和ZCuSn5Pb5Zn5阴极极化电流密度均显著增加。根据混合电位理论可知,偶对电偶腐蚀速率将显著增加,说明在流动海水环境中,电偶腐蚀速率是由20#钢阳极反应动力学和ZCuSn5Pb5Zn5合金阴极反应动力学共同控制的。当海水流速从1 m/s增大至3 m/s后,腐蚀初期,流速对20#钢阳极反应动力学基本无影响,但ZCuSn5Pb5Zn5阴极反应速率随着流速增加而小幅度增加,进而导致电偶腐蚀速率小幅度增加,说明海水流速从1 m/s增加到3 m/s后,电偶腐蚀速率增加初期主要是由于ZCuSn5Pb5Zn5合金阴极反应速率增加导致;连续冲刷后,20#钢表面沉积有大量的腐蚀产物,但流速越高,海水剪切力越大,腐蚀产物越不容易在20#钢表面沉积,因此后期20#钢在不同流速条件下阳极极化动力学不同,海水流速从1 m/s增加至3 m/s,后期电偶腐蚀速率受20#钢阳极反应动力学和ZCuSn5Pb5Zn5阴极反应动力学共同影响。当海水流速继续增大至5 m/s后,剪切力增加,导致疏松性腐蚀产物剥落,20#钢表面形成了致密性较高、活性低的腐蚀产物沉积层,沉积层防护性能有所提高,导致电偶腐蚀速率略有下降。图12为通过以上分析得到偶对在不同流速以及不同冲刷时期电偶腐蚀机理。
图11
图11
实验材料在不同流速海水中的极化曲线
Fig.11
Polarization curves of two test materials in seawater with different flow rates
图12
图12
20#钢/ZCuSn5Pb5Zn5合金偶在海水中的电偶腐蚀机理图
Fig.12
Mechanism diagrams of galvanic corrosion of 20# steel/ZCuSn5Pb5Zn5 couple in seawater
4 结论
(1) 20#钢与锡青铜偶对在流动海水条件下的电偶腐蚀速率远远大于静态条件下的。海水流速为1 m/s时,其电偶腐蚀电流密度是静态条件下的17.5倍;随着流速继续增大,电偶腐蚀速率先升高后小幅度下降。
(2) 静态以及低海水流速下,20#钢/锡青铜电偶腐蚀速率是由20#钢阳极反应动力学和ZCuSn5Pb5Zn5阴极反应动力学共同控制;当流速达到3 m/s后,20#钢/锡青铜电偶腐蚀速率在冲刷初期受ZCuSn5Pb5Zn5阴极反应过程控制,而在腐蚀中后期受20#钢阳极反应动力学和ZCuSn5Pb5Zn5阴极反应动力学共同控制。
参考文献
Review of research on the environmental corrosion of ship seawater systems
[J].
船舶海水管系的环境腐蚀研究进展
[J].
Study of naval vessel galvanic corrosion
[J].
舰船材料的电偶腐蚀研究
[J].
Control of galvanic corrosion of dissimilar metals of seawater pipelines in naval vessels
[J].
舰船海水管系异金属电偶腐蚀的控制
[J].
Study on galvanic corrosion of steel couples in seawater
[J].
海水中钢的电偶腐蚀研究
[J].
Research progress on the long period corrosion law of metallic materials in seawater
[J].
金属材料长周期海水腐蚀规律研究
[J].
Analysis of corrosion failure of bronze cut-off valve in marine seawater pipe system
[J].
船舶海水管路青铜截止阀腐蚀失效分析
[J].
Copper-nickel alloy for the construction of ship and boat hulls
[J].
Galvanic corrosion of carbon steel/titanium and carbon steel/titanium/navel brass in seawater
[J].
碳钢/Ti和碳钢/Ti/海军黄铜在海水中电偶腐蚀的研究
[J].
Recent advances and prospect of galvanic corrosion in marine environment
[J].
海洋环境电偶腐蚀研究现状与展望
[J].
Progress in research on factors influencing galvanic corrosion behavior
[J].
电偶腐蚀影响因素研究进展
[J].
Galvanic corrosion of magnesium alloy AZ91D in contact with an aluminium alloy, steel and zinc
[J].
Progress of study on Erosion-corrosion
[J].
冲刷腐蚀的研究现状
[J].
Development of synergy model for erosion-corrosion of carbon steel in a slurry pot
[J].
Progress in research methods for erosion-corrosion
[J].
冲刷腐蚀研究方法进展
[J].
Corrosion behaviour of metallic materials in high velocity seawater
[J].
高流速海水中金属材料的腐蚀行为
[J].
Kinetic law of galvanic corrosion in flowing seawater
[J].
流动海水中电偶腐蚀动力学规律
[J].
Galvanic Corrosion behavior of B10/H62 couple in Seawater of different flowing rate
[J].
不同流速海水中B10/H62电偶腐蚀规律
[J].
Effect of flowing rate on galvanic corrosion associated with 316L/2205 and 431/2205 stainless steel couples in 3.5%NaCl solution
[J].
流动对316L/2205和431/2205不锈钢在3.5%NaCl溶液中电偶腐蚀的影响
[J].
Effect of corrosive media on galvanic corrosion of complicated tri-metallic couples of 2024 Al Alloy/Q235 Mild Steel/304 Stainless Steel
[J].Galvanic corrosion of tri-metallic couples is more complicated than that of bi-metallic couples. In this study, the effect of the pH of corrosive media on the galvanic corrosion of 2024 Al alloy/Q235 mild steel/304 stainless steel tri-metallic couples was investigated using potentiodynamic polarization, scanning electron microscopy, scanning vibrating electrode technique and a multi-channel galvanic corrosion meter. The results show that 2024 always acts as the only anode in 3.5 wt% NaCl at pH 5.56, 9.72 and 12.0, while both Q235 and 2024 act as anodes at pH 2.39 in the initial stage and then the role of Q235 changes at longer coupling time, which can be attributed to the effect of pH on the surface film of 2024. It is also found that the galvanic current density of a tri-metallic couple is the superposition of two bi-metallic couples when cathodic reactions are controlled by the diffusion of oxygen, otherwise it is smaller than that of the sum of two bi-metallic couples. The localized corrosion instead of uniform corrosion of anodic metal is accelerated by galvanic corrosion.
Investigation on galvanic corrosion behaviors of CFRPs and Aluminum Alloys systems for automotive applications
[J].
Galvanic corrosion in the systems Titanium/316 L Stainless Steel/Al Brass in Arabian gulf water
[J].
Corrosion behavior of H62 brass alloy/TC4 titanium alloy welded specimens
[J].
H62黄铜/TC4钛合金焊接件腐蚀行为研究
[J].
Study on the deterioration processes of organic coating/low alloy steel systems in simulated deep-sea environment
[D].
模拟深海环境下有机涂层/低合金钢体系失效过程的研究
[D].
Corrosion resistance and antifouling performance of copper-bearing low-carbon steel in marine environment
[J].
含铜低碳钢在海洋环境下的耐蚀和防污性能的研究
[J].
Corrosion behavior of Q235 Steels in atmosphere at Deyang district for one year
[J].
Q235钢在德阳大气环境中腐蚀行为研究
[J].